|
Report
on a trial to study the use of Vitex agnus castus extract
on cases of Equine Cushing's Disease.
Protocol
The trial was conducted between November 2001 and March 2004. 120 horses and ponies
are included in the study. All animals showed clinical signs of Equine Cushing's
Disease (ECD) and had blood concentrations of endogenous ACTH (eACTH), insulin,
cortisol and glucose measured before joining the trial. Those with eACTH concentrations
of greater than 50 pg/ml were eligible for inclusion in the trial. Animals were
given Vitex at the following administration rates;. 40 ml per day for animals
less than 800lbs, 60 ml per day for those between 800 lbs and 1150 lbs and 80
ml per day for those over 1150 lbs. No other alterations in the usual management
was instituted and no other drugs were given to the animals during the trial period.
The owners were required to submit quarterly reports as to the animal's clinical
condition, noting the following parameters; occurrence of hair loss, diet, daily
water consumption, weight (using a weightape), occurrence of laminitis or other
medical problems. In addition photographs of the animal were also requested to
accompany each report. At the end of the 12 month trial period a final report
was required and a retest to measure eACTH, insulin, cortisol and glucose.
Preliminary Results
Twenty five animals completed the year of the trial. Their owners submitted full
reports including the final blood test results. This group consisted of 13 mares
and 12 geldings whose ages ranged from 14 to 26 years. The amount of Vitex was
increased in four animals which had shown no alteration in demeanour or clinical
signs after 3 months at the recommended administration rate. None of these received
more than double the original amount.
Clinical signs
All owners reported improvement in the demeanour of the animals, they became more
active, less depressed and the term rejuvenated was often used by owners. 22 owners
reported that the animals shed their thick coats during the early part of the
trial period and the next year's coat was more normal in appearance. 20 animals
were reported to have shown reduced polydipsia (excessive drinking) and hyperhidrosis
(sweating). Two of the 25 animals suffered one or more episodes of acute laminitis
during the trial period. One horse which had been dangerously aggressive prior
to starting the trial returned to his normal demeanour during the Vitex trial.
Laboratory results
12 animals showed a reduction in eACTH during the period of the trial though only
2 animals had final concentrations of less than 30 pg/ml. 9 animals showed an
increase in eACTH and the concentration in 4 animals did not alter during the
trial.
9 animals showed a reduction
in plasma insulin concentration during the period of the trial, 7 of which had
concentrations of less than 50 UIU / ml in their final samples. 13 animals showed
an increase in plasma insulin while the concentration in 3 animals did not alter
over the 12 months.
13 animals showed a reduction
in plasma cortisol over the trial period to less than 155 nmol/L in their final
sample. 10 animals showed a rise in cortisol and the plasma cortisol did not alter
in 2 animals during the trial period.
Discussion
The majority of the 25 horses for which complete trial data were available were
chronic founder cases which had prior to joining the trial suffered recurrent
episodes of acute laminitis. Only two cases suffered from laminitis during the
12 month trial period. The horses which did not survive the period of the trial
were euthanased or died due to a variety of serious systemic diseases which commonly
affect geriatric equines with ECD. In many cases inclusion in the trial was a
"last ditch" attempt which these owners were prepared to undertake.
There appears to be no trend towards normalisation of plasma concentrations of
eACTH, insulin or cortisol during the trial period, which may suggest that the
changes observed in the animals are not be related to the effects of any of these
parameters. The most consistent finding reported by the owners was the lifting
of the depression which all of these cases showed, they became more energetic,
interested in their surroundings and appeared to enjoy life again.
|
Case 4
Before Vitex
Hyperhidrosis (excessive sweating) was a constant clinical sign. This horse had
also become aggressive.
|
Case 4
After Vitex
The hyperhidrosis has resolved and his demeanour returned to normal
|
|
|
|
| |
|
|
Case 7
Before Vitex
showing
hirsutism (abnormally long coat) and depression
|
Case 7
After Vitex treatment;
hirsutism and depression have resolved
|
 |
 |
 |
 |
| |
|
|
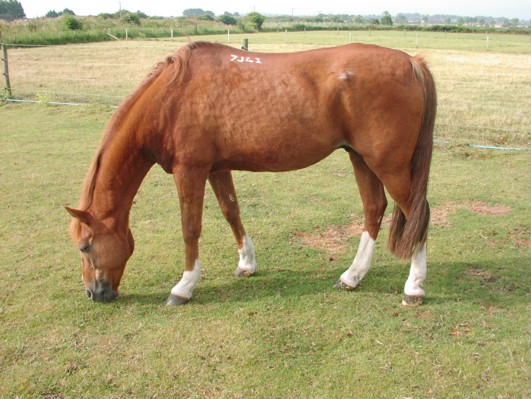
Case 21.
Before Vitex
showing hirsutism, depressed attitude and pot bellied appearance
|
Case 21.
After Vitex
clinical appearance and attitude are much improved
|
 |
 |
|








